 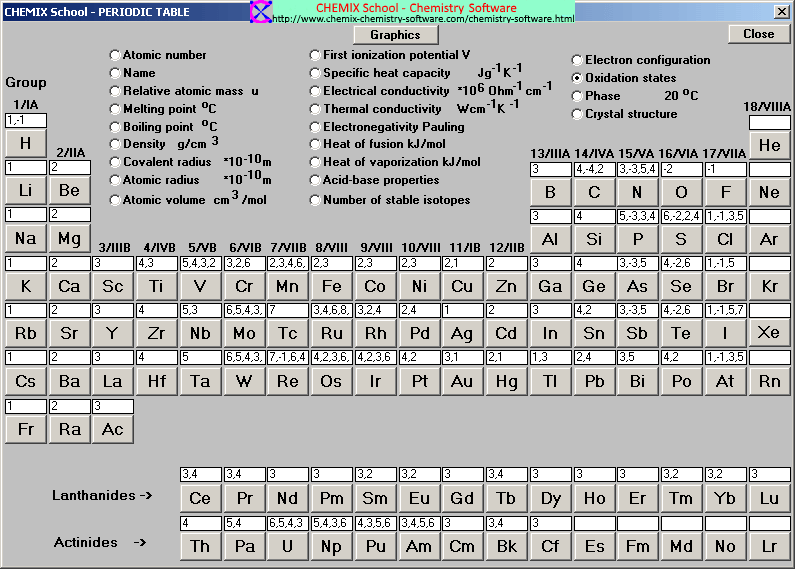
The second way for an atom to obtain an octet of electrons is by sharing electrons with another atom. The resulting compounds are called ionic compounds. Because opposite charges attract (while like charges repel), these oppositely charged ions attract each other, forming ionic bonds. If an atom has lost one or more electrons, it is positively charged and is called a cation. If an atom has gained one or more electrons, it is negatively charged and is called an anion. Recall that atoms carrying positive or negative charges are called ions. Those that lose electrons become positively charged, and those that gain electrons become negatively charged. Because some atoms will lose electrons and some atoms will gain electrons, there is no overall change in the number of electrons, but with the transfer of electrons the individual atoms acquire a nonzero electric charge. One way is the transfer of electrons between two atoms until both atoms have octets. There are two ways for an atom that does not have an octet of valence electrons to obtain an octet in its outer shell. The tendency of an atom toward a configuration in which it possesses eight valence electrons is referred to as the “ Octet Rule.” In these lower energy states, the outermost energy level has eight electrons (an “octet”). This instability drives them toward the lower energy states represented by the noble gases that are nearby in the periodic table. The elements in the other groups have subshells that are not full, so they are unstable when compared to the noble gases. They are already at a low energy state, so they tend to stay as they are. These elements have electron configurations characterized by full s and p subshells. For atoms, these lower energy states are represented by the noble gas elements. Lower energy configurations are more stable, so things are naturally drawn toward them. Throughout nature, things that are high in energy tend to move toward lower energy states. Deviations from this ratio result in charged particles called ions. This one-to-one ratio of charges is not, however, the most common state for many elements. The overall charge on the atom is zero, because the magnitude of the negative charge is the same as the magnitude of the positive charge. This is because the number of electrons (negative in charge) is equal to the number of protons (positive in charge). Up until now we have been discussing only the elemental forms of atoms which are neutrally charged. 3.1 Introduction to the Octet Rule 3.2 Ions and the Periodic Table Common Cations Common Anions Ions of Transition Metals 3.3 Ionic Bonding 3.4 Practice Writing Correct Ionic Formulas 3.5 Naming Ions and Ionic Compounds 3.6 Polyatomic Ions 3.7 Naming Polyatomic Ions 3.8 Properties and Types of Ionic Compounds 3.9 Arrhenius Acids and Bases 3.10 Focus on the Environment – Acid Rain 3.11 Chapter Summary 3.12 References This text is published under creative commons licensing, for referencing and adaptation, please click here. Oxygen will gain 2 electrons.CH104: Chapter 3 – Ions and Ionic Compounds The second shell has six electrons ( 2 s 22 p 4) and needs two electrons to achieve octet. The electron configuration of O atom is 1 s 22 s 22 p 4. How many electrons must O lose/gain to achieve octet? Write the formula of the resulting ion and its electron configuration. Write the electron configuration of oxygen atom (Z=8).  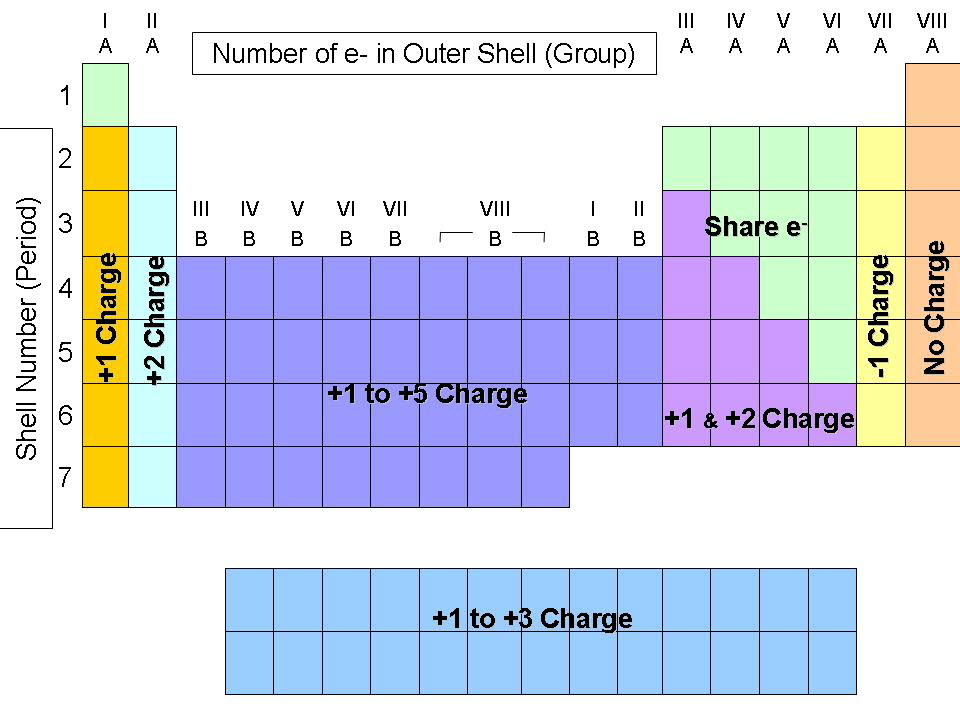
In macroscopic samples of sodium chloride, there are billions and billions of sodium and chloride ions, although there is always the same number of cations and anions. 
The number of electrons lost by the sodium atom (one) equals the number of electrons gained by the chlorine atom (one), so the compound is electrically neutral. Notice that there are no leftover electrons. The resulting combination is the compound sodium chloride. With two oppositely charged ions, there is an electrostatic attraction between them because opposite charges attract. On the right, the chloride ion has 18 electrons and has a 1− charge. On the left, the chlorine atom has 17 electrons. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
